At present, the need of high-endurance new energy vehicles forces the energy density of batteries to become higher and higher, and the use of thick electrodes with high load density active materials is one of the most practical strategies. However, their long cycle use process is accompanied by serious attenuation of electrochemical performance, power performance is not satisfied, and the capacity retention rate is getting worse and worse. So what exactly is causing the bottleneck of poor performance?
Kyu-Young Park et al. explored the key processes that restrict battery decay by designing thick electrodes with different area degrees.
1. Experimental design
Using NCM622: carbon black: PVDF 97:1.5:1.5 ratio and NMP mixed into pulp, after coating, drying and roller pressing, two kinds of electrodynamic half cells (2032) with different surface densities (20 and 28mg/cm-2) were prepared, and the pressure was between 2.8 and 2.9, in order to ensure better porosity. The charge and discharge cycle of the multi-channel device was carried out with the charge and discharge interval of 2.8-4.3V and the rate 1C was about 150mA/g. EIS, chemical composition and morphology were analyzed after every 20 cycles.
2. Results and discussion
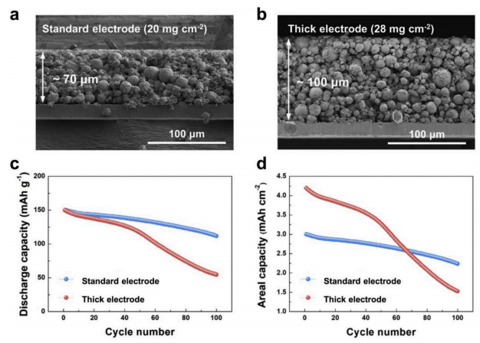
The following is the cross-section diagram of the electrodes of two thickths, respectively 70 and 100μm(standard electrode, thick electrode), the rest of the porosity, 1C current density and other design parameters are basically the same, and then the 1C cycle test is carried out. It is found in Figure c that although the capacity of the thick electrode of 100μm is only 40% higher than that of 70μm, but after 100 battery cycles, The thick electrode has a capacity retention rate of only 36%, while the standard electrode has a capacity retention rate of 76%. Even taking into account the volume specific capacity, the thick electrode after attenuation in Figure c is still much lower than the electrode. Interestingly, in Figure c, even in the initial cycle process, the circulation curves of the thick electrode and the standard electrode are close, and the attenuation degree is similar. Thick electrodes are getting worse.
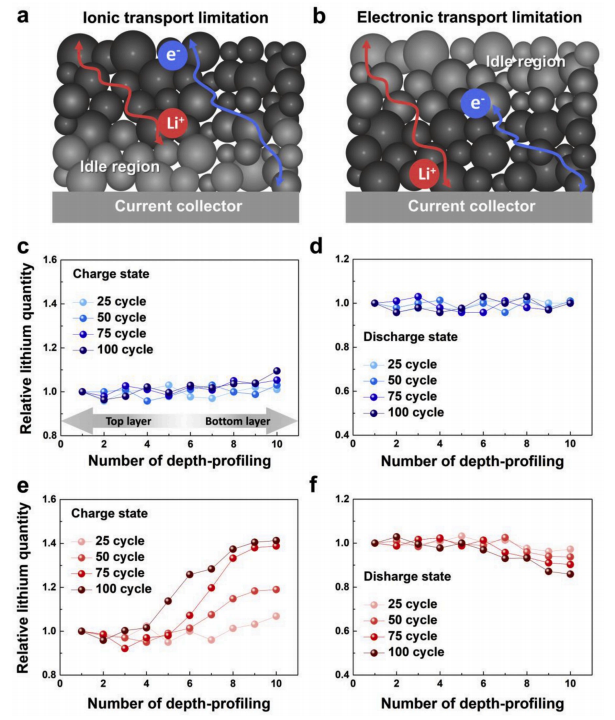
In illustrating the poor electrochemical performance observed, the authors note that thick electrodes may be subject to kinetic limitations caused by how fast or slow charge carriers migrate, which in electrochemical processes is either controlled by lithium-ion transport or by the transport of electrons that accumulate along the electrode. And, in each case, assuming that the main source of supply of electrons and lithium ions to the electrode is carried out from the electrode/collector interface and the electrode/electrolyte interface, in each case there will be a clear spatial distribution of both after the reaction.
3. Conclusion
By using batteries designed with different electrode thickness, the authors verify that lithium ion diffusion is the limiting factor of charge transfer, but not electron transfer. This is also the reason why SOC at different locations is uneven, voltage drop IR increases, particle breakage and even battery diving under charge and discharge in batteries designed with thick electrodes. The electrode plate is designed according to the ion transport characteristics to avoid the phenomenon of excessive local current density, so as to improve the battery life